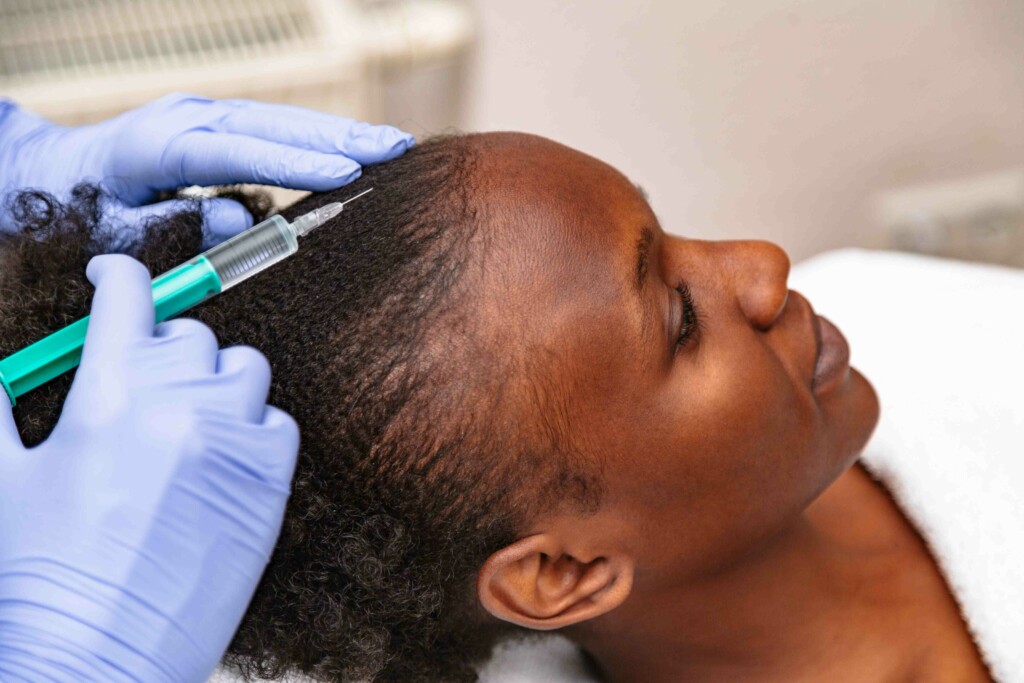
In June 2022, the first systemic treatment for alopecia areata — Olumiant (also known as Baricitinib) — was approved by the U.S. Food and Drug Administration. Since then, several other Janus kinase (JAK) inhibitors have been in clinical trials to treat multiple types of alopecia, including alopecia areata and cicatricial alopecia. (1)
The next most likely candidate to be approved for hair loss is Ritlecitinib, also referred to PF-06651600. This Pfizer-owned drug is being trialed for the treatment of alopecia areata in adults, adolescents, and children between the ages of 6 and 12. It’s also being tested in adults with cicatricial alopecia (a form of scarring hair loss). (2-4)
Ritlecitinib will likely first be approved for the treatment of alopecia areata in adolescents over 12 and adults sometime in 2023. This drug has not yet been approved for any other indications.
What Is Ritlecitinib?
Ritlecitinib, a JAK inhibitor drug, is primarily being tested for the treatment of alopecia areata. Phase 2 and 3 clinical trials are currently being conducted in adults and adolescents over 12 years of age. And more recently, a Phase 1 clinical trial was listed testing the use of Ritlecitinib in children between 6 and 12 years old. (1-4)
There are currently 13 clinical trials for Ritlecitinib listed on ClinicalTrials.gov. Six of these trials have been completed, though only one of these completed trials focused on the treatment of alopecia areata. There are currently seven ongoing trials for the treatment of alopecia areata, cicatricial alopecia, celiac disease, rheumatoid arthritis, and vitiligo. Ritlecitinib is a Pfizer product, which means that all of these clinical trials are sponsored by Pfizer. (1)
What Is Alopecia Areata?
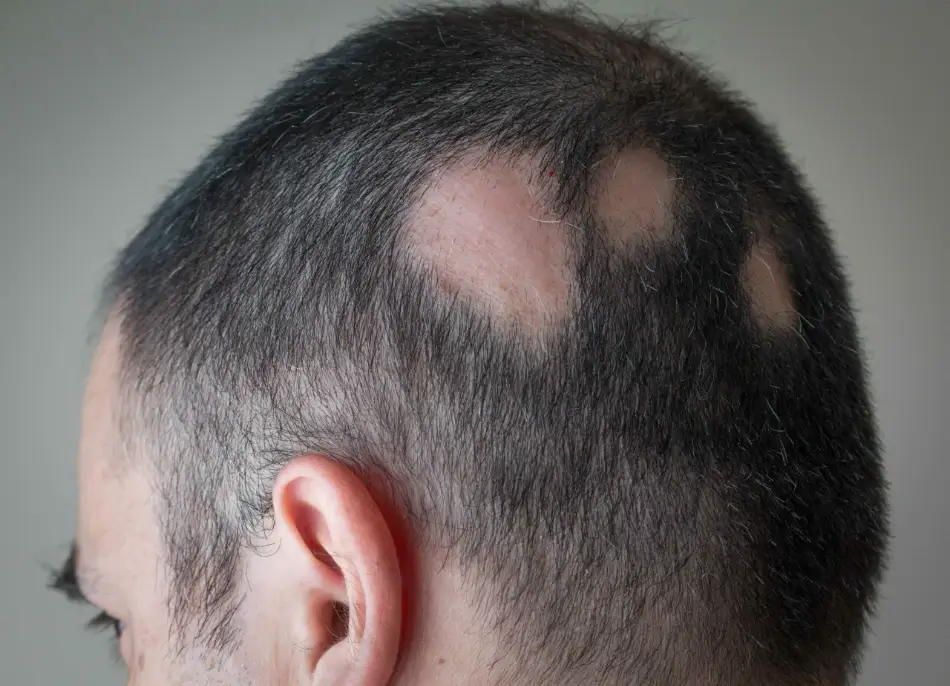
Alopecia areata is an autoimmune form of hair loss that typically causes round patches of hair loss on the scalp. In some cases, alopecia areata can be more extensive, affecting the entire scalp, or multiple parts of the body, including body hair and eyelashes, eyebrows, and other facial hair. This form of hair loss is thought to affect about 147 million people worldwide. (5,6)
What Is a JAK Inhibitor?
JAK inhibitors are drugs that work by blocking proteins released by the immune system. These proteins, known as cytokines, can activate the Janus kinase pathway, resulting in inflammatory responses that can be detrimental to both skin and hair. When cytokines are prevented from activating this pathway, inflammation is also prevented, which in turn supports the regrowth of healthy hair. JAK inhibitor drugs essentially work by suppressing overactive autoimmune responses. (5,6)
Ritlecitinib Dosing for Alopecia
In current studies, the dosing of Ritlecitinib for alopecia areata can range between 10 milligrams and 200 milligrams. People may start on a dose of 200 milligrams for the first month, after which the dose is usually reduced to 30 milligrams or 50 milligrams. In all cases, this drug is meant to be taken once a day. (1-4)
Ritlecitinib for children is still highly exploratory, with the first clinical trials testing just a 7-day period of a daily 20-milligram dose. Ritlecitinib has also been tested in smaller doses of 10 milligrams in healthy adults. Future studies may determine if such low doses are appropriate for maintenance control for chronic conditions. (1-4)
One study is also exploring the use of Ritlecitinib for cicatricial alopecia. Dosing has been set higher for this type of scarring alopecia, with a month-long 200-milligram starting dose followed by a daily dose of 100 milligrams. (1-4)
Are There Other JAK Inhibitors for Hair Loss?
Compared to minoxidil and other long-standing hair loss treatments, JAK inhibitors are comparatively new medications. But Ritlecitinib is not the first JAK inhibitor to be trialed in people with alopecia areata.
In June 2022, the FDA approved an oral medication called Olumiant (also known as Baricitinib) for the treatment of severe alopecia areata. This JAK inhibitor was developed by Eli Lilly. (1)
Olumiant was the first FDA-approved systemic treatment for alopecia areata. It was originally approved for the treatment of some adults with moderate-to-severe active rheumatoid arthritis in 2018. It can also be used to treat COVID-19 in certain hospitalized adults. (1,8)
There’s also another JAK inhibitor, Deucravacitinib, being tested in alopecia areata trials. Deucravacitinib, a Bristol-Myers Squibb product, is already approved for the treatment of moderate to severe plaque psoriasis. However, when it comes to hair loss, this JAK inhibitor is trailing behind. It only started phase 2 trials at the end of 2022, which are scheduled to finish at the end of 2024. (9,10)
Other JAK Inhibitors in Dermatology
In the last few years, Janus kinase (JAK) inhibitors have made major breakthroughs in dermatology. It all kicked off when the U.S. Food and Drug Administration approved Opzelura cream, (also known as Ruxolitinib) to treat atopic dermatitis and shortly after, vitiligo. Since then, the FDA has also approved (11):
- CIBINQO® (also known as Abrocitinib) for moderate to severe atopic dermatitis
- RINVOQ® (also known as Upadacitinib) for moderate to severe atopic dermatitis
- SOTYKTU® (also known as Deucravacitinib) for moderate to severe plaque psoriasis
- XELIJANZ® (also known as Tofacitinib) for symptomatic psoriatic arthritis
Is Ritlecitinib FDA Approval Coming Soon?
The U.S. Food and Drug Administration and European Medicines Agency accepted the New Drug Application for Ritlecitinib in September 2022. Pfizer has also completed regulatory submissions for this medication in China, Japan, and the United Kingdom. However, these submissions have been made for use of the drug in adults and adolescents 12 and older. (7)
Is There a Forecasted Ritlecitinib Release Date?
There is currently no exact forecasted release date for Ritlecitinib. However, a decision from the FDA is forecasted to arrive in mid-2023. A decision from the EMA is expected before the year’s end. (7)