The latest pattern hair loss clinical trial to be announced this year is a laser hair therapy study being conducted by StimuSIL, a European-American start-up. This study is testing the safety and effectiveness of a novel, patented laser and microneedling device on men with androgenetic alopecia.
StimuSIL’s SAGA-001 Clinical Trial
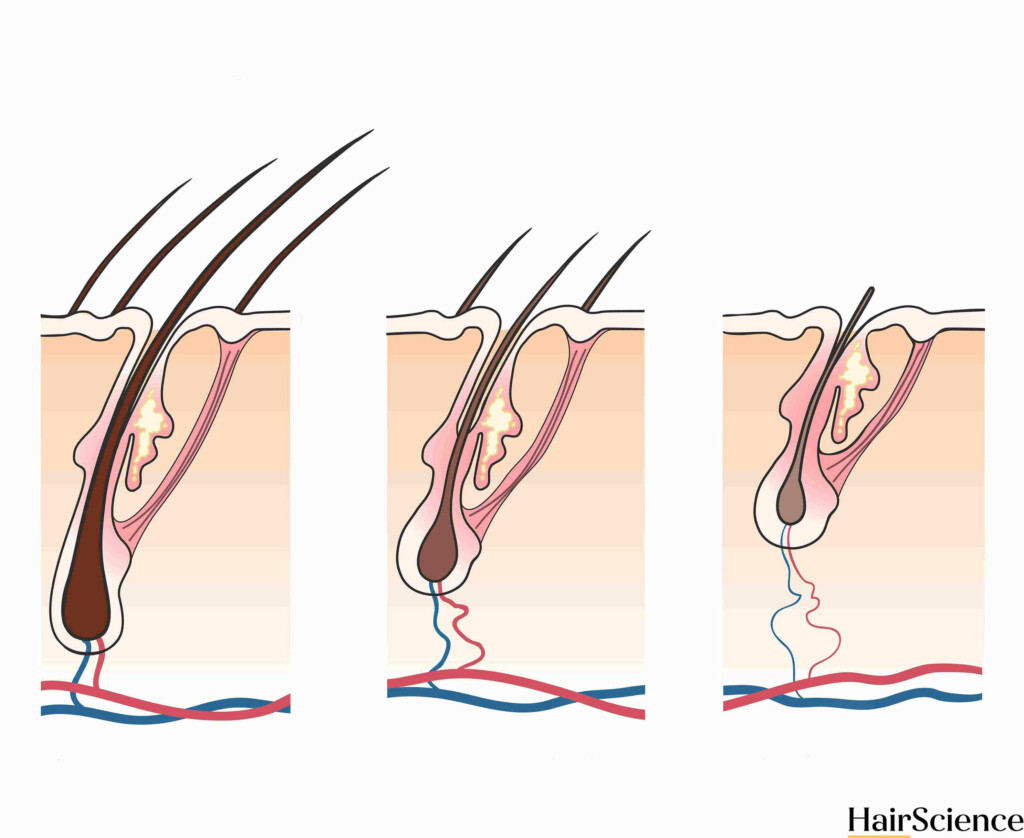
StimuSIL is a biotech startup that is developing medical devices to treat dermatological and aesthetic issues, including hair loss. According to their website, they are focused on photobiomodulation, which is also commonly referred to as low-level laser therapy or LLLT. Laser hair therapy uses laser light to trigger a series of cellular events that can positively affect skin and hair follicles. This cellular cascade increases levels of certain enzymes and chemicals, improving blood flow to the scalp and reactivating dormant hair follicles. (1-6)
StimuSIL’s current study is testing a novel laser and microneedling device that they refer to as SAGA-001. They are testing two types of treatments: SAGA–001 A and SAGA-001 B. These SAGA-001 devices seem to be almost identical, except for the additional metal microneedles present in device A. This seems to be the first androgenetic alopecia clinical trial to combine microneedling with laser hair therapy. (3)
SAGA-001 vs. LLLT for Hair Loss
StimuSIL hasn’t revealed much about the SAGA-001, other than that it uses both lasers and microneedles. Their clinical trial listing also mentions that they are testing two versions of this medical device. It seems that they are trying to determine the ideal regime for stimulation and microwounding – specifically, microneedling – when treating pattern hair loss. (3)
It is unclear what kind of laser is being applied to the scalp during SAGA-001 treatments. However, an article on StimuSIL’s website says that they created a modified, more effective version of low-level laser therapy. (2,3)
Their device appears to deliver minimally invasive long-wavelength light, as is used in traditional laser hair therapy. But StimuSIL has apparently changed how the light is delivered and patented its novel approach. (2,3)
According to the article, “StimuSIL’s precision system allows light to reach the hair follicle directly. This allows the StimuSIL device, SAGA-001, to deliver 5 to 6 times as much power to produce the maximum possible beneficial effect without heating or burning the skin.”
Interestingly, changing the delivery of the laser and its power means that the SAGA-001 device will only be available to clinicians and other medical professionals. This makes it different from the vast majority of LLLT devices for hair loss, like the Theradome, iRestore, or Hairmax Laser Cap, which are all sold directly to consumers for home use. (2)
SAGA-001 Treatments
SAGA-001 treatments take around 20 to 30 minutes. They are designed to be applied by a trained medical professional. These clinic-based “lunchtime procedures” are required relatively infrequently.
Unlike most other hair loss treatments, which need to be used on a daily basis, StimuSIL’s treatment only needs to be performed once every two weeks. This makes SAGA-001 treatments more comparable to other clinic-based hair loss treatments, like platelet-rich plasma (PRP) therapy. PRP treatments, which use high concentrations of platelets to stimulate dermal papilla cells and promote hair growth, are also applied every few weeks or once a month by a trained medical professional. (2,3,7)
Given the duration of their study, it seems like StimuSIL expects people with androgenetic alopecia to need biweekly treatments for about 24 weeks. It also seems that the SAGA-001 can be used as a stand-alone hair loss treatment, as in this clinical trial, or alongside existing treatments, like minoxidil or topical DHT blockers. (2,3)
“Depending on the patient, doctors may recommend combining SAGA-001 treatments with minoxidil, newer topicals like KX-826 and Breezula, or even as a complement to enhance the results of hair transplants,” Pablo Villalba, one of StimuSIL’s founders, said in an interview. “Eventually, we’d like everyone to be able to customize their hair loss treatment to maximize effectiveness and results.” (2,3)
StimuSIL reported recruiting their first subject on May 31, 2023, and is still currently recruiting participants for its clinical trial. Given the duration of their study, they may only have results to report during Q3 2024. (4)
Interested in Joining StimuSIL’s SAGA-001 Study?
If you are interested in joining the SAGA-001 clinical trial, you’ll have to be male, between 22 and 55 years of age, and have mild-to-moderate androgenetic alopecia (classified between Norwood-Hamilton stages IIa and V).
Participants must also have Fitzpatrick Skin Types I-IV. This is a common restriction with LLLT treatments as phototherapies are not always safe or effective for people with more melanin in their skin. (3,8,9)
StimuSIL’s list of exclusionary criteria is quite extensive. If any of the following apply, you won’t be able to take part in the study: (3)
- Prior use of other hair loss treatments (in the last 6 months)
- Prior or current use of medications or products that can influence hair loss or hair growth
- Use of semi-permanent hair products in the last 30 days
- Other health issues (besides androgenetic alopecia), such as bleeding disorders, autoimmune issues, keloids, hepatic or renal disease, epilepsy, scalp infections, and chronic skin issues affecting the scalp.
- Other types of hair loss (besides androgenetic alopecia), such as alopecia areata, telogen effluvium, anagen effluvium, and acquired cicatricial alopecia.
Additionally, people who have undergone a hair transplant, scalp surgery, scalp radiation, or chemotherapy are not able to participate in this study.
If you are a reasonably healthy individual and can make regular visits to StimuSIL’s clinical trial sites in Turkey, you are likely eligible for this clinical trial. If you’d like to participate in this study, reach out to ana@stimusil.com or one of the investigators running their clinical trial.
If you’re not based in Turkey but would still like to participate in a future clinical trial, reach out to StimuSIL directly at accounts@stimusil.com. According to Pablo Villalba, StimuSIL intends to conduct studies in North America and Europe in the near future, and has a waitlist for people interested in participating in future trials.
You may also want to look into the new fractional non-ablative clinical trial, which is exploring the combination of lasers and microwounding using only light (no physical microneedles are used). This study is currently accepting participants with both androgenetic alopecia and scarring alopecia.