Androgenic alopecia is the most common form of hair loss, affecting 80 percent of Caucasian men and up to 50 percent of Caucasian women. It’s also known as pattern hair loss.
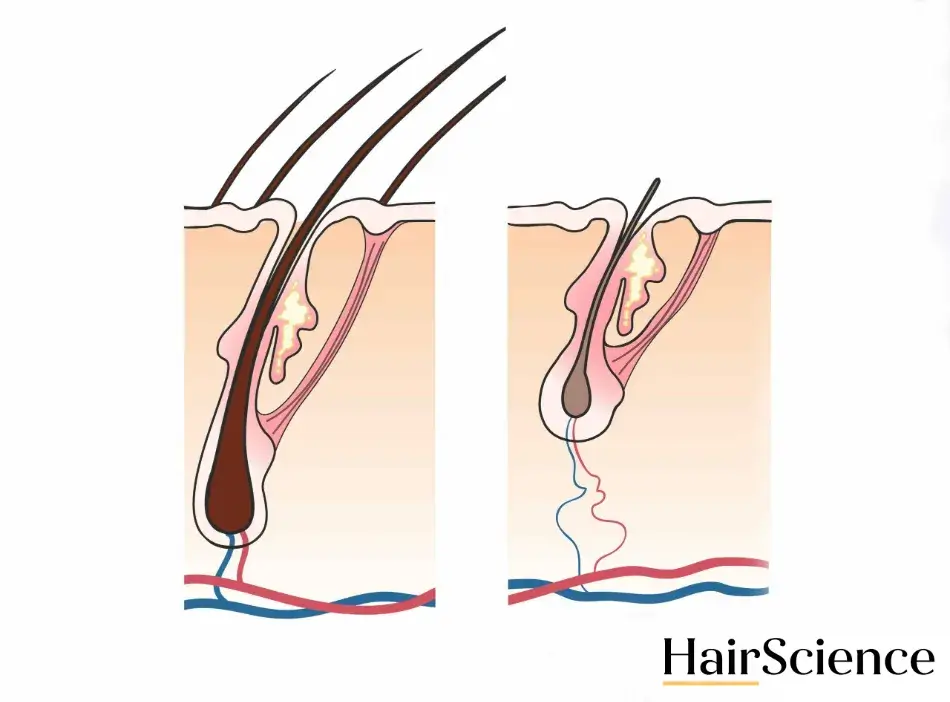
Androgens are naturally occurring hormones that play an important part in the development of male characteristics and sexual traits. They’re present in both men and women. In most cases androgens are helpful, but in some people, an androgen called dihydrotestosterone (DHT) can bind to hair follicles in excess.
DHT binding shrinks hair follicles, which is called miniaturization. The miniaturization process shortens the hair growth cycle and causes thinner, weaker strands of hair to grow. Over time this can stop hair growth altogether.
There are three FDA-approved hair loss treatments for androgenic alopecia. These include a topical treatment called minoxidil, an oral treatment called finasteride, and low-level laser therapy devices. There are also a range of promising new treatments currently going through clinical trials. One of these is Breezula®, a medicine that works as an androgen receptor inhibitor. (1)
What Is Breezula®?
Breezula® is an androgen receptor inhibitor that’s being tested as a topical treatment for male and female androgenic alopecia. It’s designed to target androgen receptors in the scalp to stop dihydrotestosterone (DHT) binding to the hair follicles. Formulations of this drug, which is known by the generic name clascoterone, are being developed by a company called Cassiopea, a part of the bigger company Cosmo Pharmaceuticals. (2,3)
Breezula® is known by a range of names including:
- Clascoterone
- CB-03-01
- Cortexolone 17α-propionate
- Winlevi®
Clacosterone cream is already available and approved at a 1 percent concentration. However, this FDA-approved medication is for the treatment of acne and is sold under the name Winlevi®. Breezula® is not yet approved as a hair loss treatment. (2,3,4)
Breezula® for hair loss is being trialed at a higher dose. Both 2.5 percent and 5 percent solutions have been trialed twice daily. A 7.5 percent solution has also been explored for use once or twice daily. Trials have shown an increase in hair growth with all percentages of the solution. The 7.5 percent solution currently seems to be the most effective. (2,3,4)
Does Breezula® Regrow Hair?
Breezula is a topical treatment applied directly to the area of the scalp affected by hair loss. It works as a novel androgen receptor inhibitor, which is thought to stop testosterone and dihydrotestosterone (DHT) from binding to hair follicles. Blocking this binding process is thought to stop the progression of androgenetic alopecia and promote the regrowth of hair. Although it’s not yet approved to treat hair loss, clinical trials showing Breezula hair results suggest it may be successful in helping people regrow hair. (2,3,4)
How Does Breezula® for Hair Loss Work?
Breezula® is a topical androgen receptor inhibitor. This drug specifically works by stopping androgens from binding to the receptors in the scalp. It works by preventing an androgen known as DHT from binding to hair follicles, which would otherwise cause symptoms of androgenic alopecia and its progression toward baldness. (2,3,4)
Both finasteride and dutasteride are 5a-reductase inhibitors – which means they’re a different type of DHT blocker. They stop testosterone being converted into dihydrotestosterone (DHT), rather than blocking androgen receptors. This lowers levels of DHT in the bloodstream, reducing the amount of DHT attaching to the hair follicles to treat androgenic alopecia. (1)
Both finasteride and dutasteride are taken orally. Finasteride is taken at a 1 milligram per day dose while dutasteride is taken at a 0.5 milligram per day dose. However, only finasteride is currently approved by the FDA as an oral medication to treat androgenic alopecia. Dutasteride is FDA-approved to treat benign prostatic hyperplasia, although it’s used as an off-label hair loss treatment and is approved for that purpose in other countries. (1)
Androgen receptors are found in many of the body’s tissues. Since both finasteride and dutasteride are taken orally, this means that these drugs can cause systemic side effects. Breezula® is applied topically and therefore, may have fewer side effects. A topical version of finasteride is available as 1 percent gel or 0.25 percent solution. While topical finasteride seems to cause fewer systemic side effects than oral finasteride, it’s not yet FDA-approved. (1)
Ketoconazole is another topical treatment that can help treat hair loss symptoms. It’s an antifungal medication that’s usually used to treat skin infections, but is used off-label to treat androgenic alopecia. Rather than being applied as a lotion, it’s usually used as a 2 percent shampoo. Unlike Breezula®, which blocks the androgen receptors, ketoconazole inhibits an enzyme that converts testosterone to DHT. In theory, it can lower levels of DHT, but results showing its ability to treat hair loss are mixed. (1)
Is Breezula® Better Than Finasteride?
Technically, finasteride is more effective than Breezula. It’s been FDA-approved for several decades, and has been proven to successfully treat androgenic alopecia in countless studies. (1,2)
Breezula® has a 7.5 percent solution that seems very promising in treating pattern hair loss. However, this medication is still going through clinical trials and is not yet approved by the FDA. (1,2)
Finasteride has positive outcomes on hair growth, with a 2022 review article showing that when taken for 24 weeks, average hair growth increased from an average of 20.2 hairs per cm2 squared to 21.1 per cm2. (5)
Initial trials for Breezula® show promise, with increased hair growth shown compared to the placebo. However, there have been no studies directly comparing the efficacy of finasteride and Breezula. But despite this, clinical trial results so far show that Breezula® could be on par with finasteride for promoting hair growth. (6)
Since finasteride is taken orally, it can have systemic side effects. These can include sexual dysfunction and mood disturbances. On the other hand, Breezula® is applied topically and trial results suggest it has very few systemic effects. (1,2,7)
A topical form of finasteride is available as a 1 percent gel or 0.25 percent solution. It’s shown to have similar effectiveness to oral finasteride. (5) Like Breezula®, topical finasteride is likely to have fewer systemic side effects than oral finasteride. It’s not yet known if Breezula® and topical finasteride are comparable in promoting hair growth.
Breezula Clinical Trial Results
Breezula® results have been vastly positive, showing increased hair growth in patients with androgenic alopecia. (2,7) A phase two, multicenter, prospective, randomized, double-blind study of men with pattern hair loss compared a variety of percentages of Breezula® to see which was the most effective. After comparing a 2.5 percent, 5.0 percent, and 7.5 percent solution, they determined that the 7.5 percent solution was the most effective. (3,8)
In this trial, 56.1 percent of patients saw increased hair growth when using a 7.5 percent solution once daily and 61.8 percent saw improvements using the 7.5 percent solution twice daily. These clinical trials also highlighted certain Breezula® side effects, which were localized to the application area. Side effects included skin irritation, redness, scaling, and dryness. However, these issues were similar when compared to a control product over a period of 12 months. (2,3)
A phase two, multicenter, prospective, randomized, double-blind trial was recently completed to test the effectiveness of Breezula® for female pattern hair loss. The results have not yet been published, but a press release reported that improvements in hair regrowth were only seen in younger women (under 30). (9)
Is There a Breezula Clinical Trial Coming Soon?
Breezula® has shown promise as a hair loss treatment, so trials are ongoing in Germany and the USA. Two Breezula® phase three trials for male pattern hair loss started at the end of June 2023 after being delayed by the COVID-19 pandemic. The trial was originally meant to be focused on the 7.5 percent dose used to treat male androgenic alopecia and run for 6 months. But instead, two identical Breezula phase three trials are now occurring simultaneously. Both are using a larger volume of 5 percent Breezula and participants will be involved in the study for between 6 to 12 months. (2,7,10)
Breezula® is not yet approved by the FDA. Results from the phase three trials will play a major role in where the product goes from here. Meetings between the company and the FDA are in progress for approval of Breezula® and more news will become available when all three trial phases are complete. (2,7)
Can You Use Winlevi® for Hair Loss?
You can’t buy Breezula® yet – but you can buy Winlevi®. Winlevi® is essentially the same drug that’s only sold in a much lower dose. It’s available as an FDA-approved topical cream that’s used to treat acne vulgaris. (3)
Although Winlevi® shares the same active ingredient as Breezula®, the formulation is different. Winlevi® is only available as a 1 percent cream compared to the 7.5 percent formula that is being trialed to treat hair loss. Therefore, it’s unlikely that Winlevi® is strong enough to help to treat hair loss. (3)
When Will Breezula Get FDA Approval?
No one – including Cassiopeia and the FDA – knows for certain of a Breezula® release date or when Breezula® FDA approval will be granted. The phase three trials need to be completed first and they have been delayed a number of times (likely due to the COVID-19 pandemic). It’s possible that within the next two to three years, approval may be granted, but this is dependent on many factors including the trial results. It’s likely that the treatment will first be approved for use in men, with the potential for approval for women to follow. (2,3)
Some drugs, such as dutasteride, are approved for other uses but not for hair loss in the USA. Yet, they are approved in other countries or sometimes used as off-label treatment options. Depending on the outcomes of the next few years, Breezula® may face a similar situation. (1)
Breezula shows great promise as a treatment for hair loss. If the FDA grants approval, it would be the only topical androgen receptor inhibitor approved to treat androgenic alopecia. It would also be the first drug with a new mechanism of action to treat androgenic alopecia in over two decades. (2)