In the last few weeks of December 2022, a new clinical trial for male androgenetic alopecia was announced. This Phase 1B/2A male pattern hair loss trial is sponsored by Dermaliq Therapeutics, Inc., an American spin-off of Novaliq GmbH, a German company that focuses on ocular therapeutics.
Dermaliq has yet to say what is in DLQ01, but it has mentioned that this topical medication is a Prostaglandin F2α analogue. DLQ01 is also known to contain Dermaliq’s proprietary hyliQ® technology, which utilizes carrier molecules that can transport solubilized active ingredients through the skin and into hair follicles. (1,2)
What We Know About the DLQ01 Clinical Trial
DLQ01 is reported to be a medication that both men and women with androgenetic alopecia can use. However, this initial randomized, double-blinded, vehicle-controlled Phase 1b/2a trial is only enrolling men. The trial is designed to evaluate the safety and effectiveness of DLQ01, additionally allowing researchers to gain an understanding of the drug’s pharmacodynamics and any systemic absorption that occurs when the medicine is applied. (1,2)
Four treatment options are being assessed in this clinical trial, all of which are meant to be applied twice a day:
- A high dose of the prostaglandin F2α analogue in its vehicle solution
- A low dose of the prostaglandin F2α analogue in its vehicle solution
- The vehicle solution used to make DLQ01, but without DLQ01’s active ingredient
- Minoxidil 5% topical solution
Dermaliq announced the randomization of its first participant on December 15, 2022. A total of 120 patients will be enrolled over the next few months. The trial, which is running at Sinclair Dermatology in Melbourne, Australia will last 24 weeks for each participant. It is expected to finish by the end of 2023. (1,2)
What Is Prostaglandin F2α?
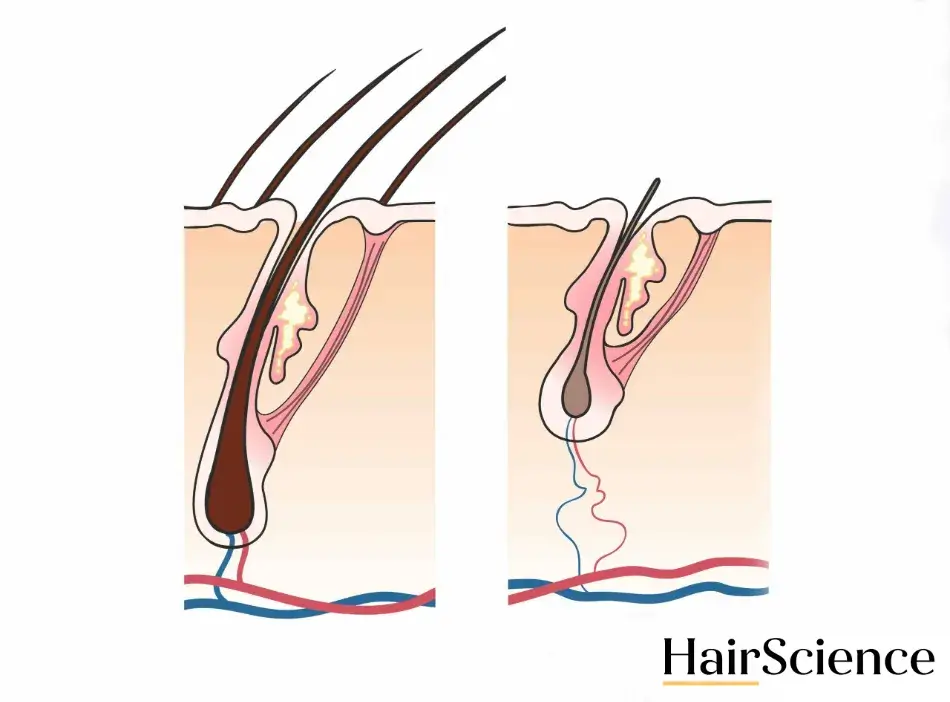
Prostaglandins are signaling molecules found in most of the human body’s tissues. Certain prostaglandins are thought to be involved in hair growth, hair loss, and the differentiation of hair follicle cells. Prostaglandin F2α is specifically thought to help promote hair growth. (3)
“Topical application of prostaglandin F2α analogue eyedrops has already been demonstrated to grow eyelashes when used to treat glaucoma,” said Prof. Rod Sinclair, MD, Professor of Dermatology at the University of Melbourne and Director of Epworth Dermatology, in a Dermaliq press release. “These agents could potentially also stimulate hair regrowth in androgenic alopecia in man and woman. However, to date, difficulties in topical delivery of this class of compounds into the hair follicle have limited their development.” Dermaliq hopes to overcome this issue by combining the prostaglandin F2α analogue with its hyliQ® technology. (2)
Interested in Joining the DLQ01 Trial?
If you have pattern hair loss, live in Australia, and are interested in this clinical trial, check out the criteria below to see if you’re eligible to join. Trial eligibility states that you must be (1):
- Male
- 18 to 60 years old
- Diagnosed with active male pattern hair loss affecting the vertex area of the scalp
- Diagnosed with a hair loss classified as Norwood-Hamilton Grades of IIIv through V, excluding IIIa, IVa, and Va grades
- Willing to maintain the same hairstyle, approximate length, and color throughout the duration of the study
- Willing to have target areas shaved
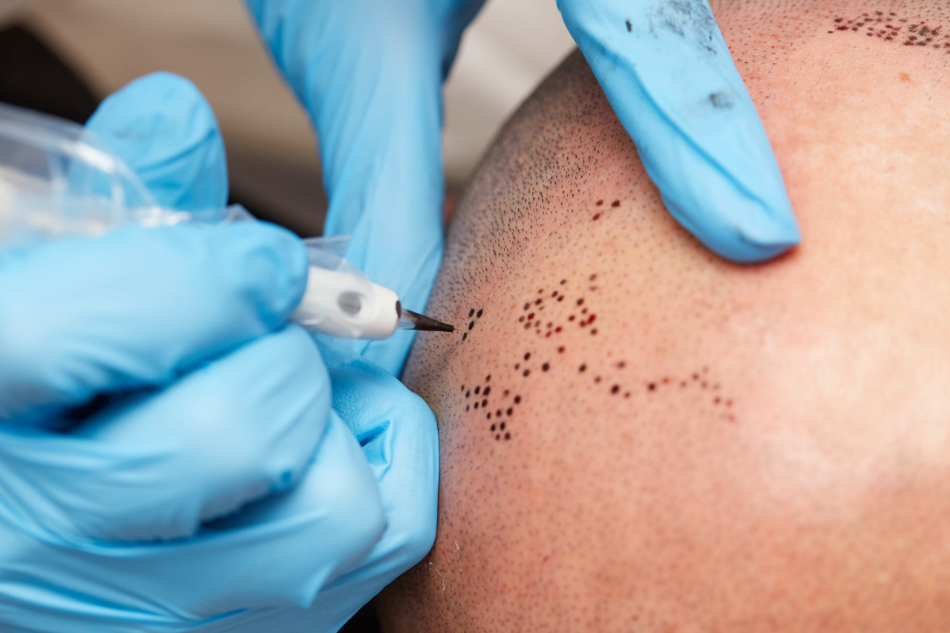
- Willing to have temporary dot tattoos placed on the scalp
The exclusion criteria state that most healthy people can apply for the study. However, those using another hair loss treatment or who have received a hair transplant are not eligible.
If you’re interested, you can register directly with Sinclair Dermatology for more information. If you’re not based in Australia and are still interested in joining a clinical trial, check out Cosmo Pharmaceutical’s phase III clinical trial for Breezula®, which started patient recruitment at the end of June 2023.